 
A comma delimited list of several species may be entered. This means that CH3 (CH2)4CH3 will be treated the same as C6H14. So we can see it has linear molecular geometry and linear electron geometry as well. Multiple specifications for an atom will be added. Thus this compound is an AX2 type molecule. It has two surrounding atoms and no lone pair. Strict number four corresponds to sp 3 hybridization.\). Molecular geometry can be determined from the VSEPR chart. The atoms at the lone pair are expected to be at 109.5 o, however, because the revulsion from the lone pair is stronger, the angle between the hydrogens is about 104.5 o: The electron geometry, therefore, is tetrahedral, and the molecular geometry is bent. The central atom has a steric number of 4 – two atoms and two lone pairs. All bonds are represented in this table as a line whether the bond is single. There are no stable AXE4, AX3E3, AX2E4 or AXE5 molecules. We are interested in only the electron densities or domains around atom A. 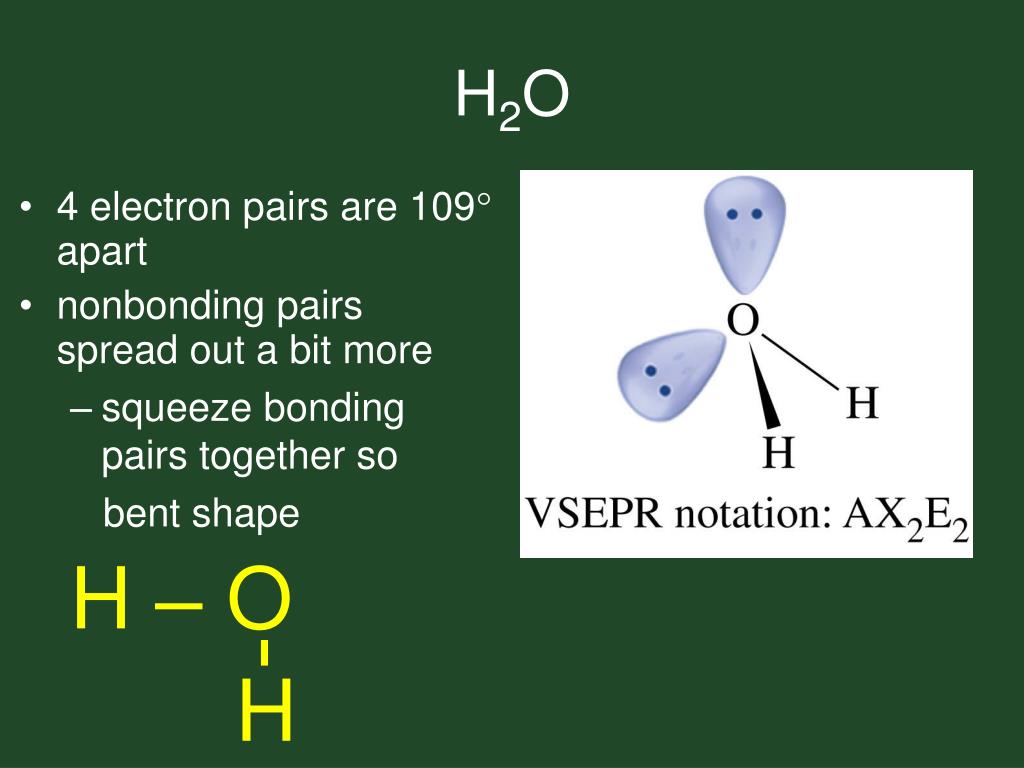
Note: There are lone pairs on X or other atoms, but we dont care. Valence electrons of Hydrogen: 12 ( as there are 2 Hydrogen atoms, we will multiply it by 2) Valence electrons of Oxygen: 6 Total number of valence electrons in H2O: 2 + 6 8 valence electrons Thus, H2O has a total of 8 valence electrons. Oxygen being more electronegative goes on the periphery and there are 6 + 2 = 8 electrons, four out of which are used to make the two bonds, and the other four go to the oxygen as lone pairs: A the central atom, X an atom bonded to A, E a lone pair on A. Electronic geometry takes into account the electron pairs that are not participating in bonding, and the electron cloud density. The molecular geometry gives water a bent shape. * Put more electronegative elements in terminal positionsĢ. Sum the valence electrons from all the atoms.ģ. Use a pair of electrons to form a bond between each pair of bound atoms.Ĥ. Add the remaining electrons to satisfy the octet for a more electronegative atom first.ĥ. If any atoms lack an octet, make a double or triple bond to give them an octet. The electronic geometry gives water a tetrahedral shape. * Hydrogen atoms are always terminal (only one bond) 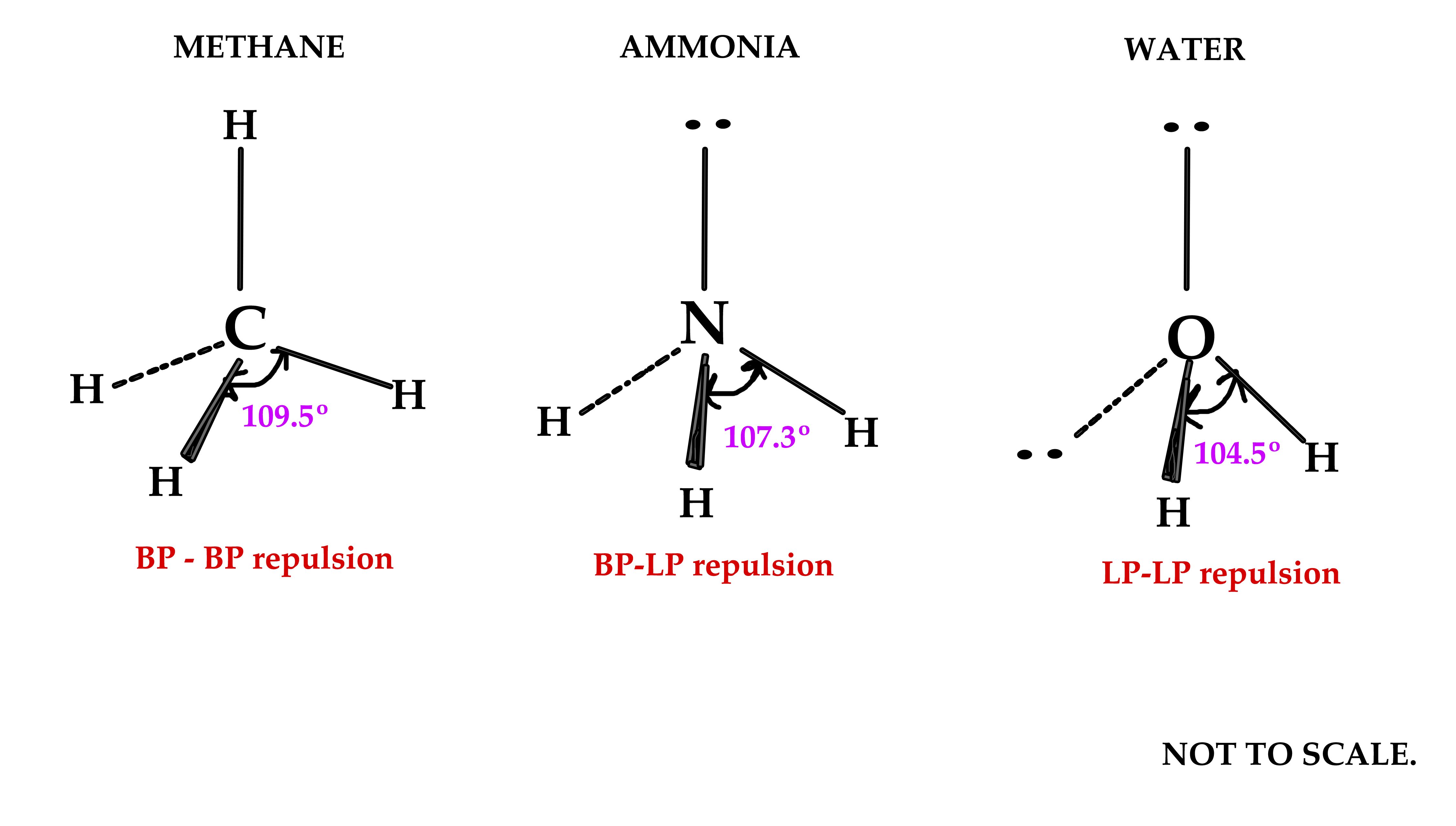
Although the bond angle should be 109.5 degrees for trigonal pyramidal molecular geometry, it decreases to 107 degrees due to the lone pair on the nitrogen atom. They are found in the atom’s outermost shell, where the force of attraction from the nucleus is the weakest. These valence electrons act as the building blocks of the structure. The electron-pair geometries shown in describe all regions where electrons are located, bonds as well as lone pairs. It is important to note that electron-pair geometry around a central atom is not the same thing as its molecular structure. Understanding the molecular structure of a compound can help determine the polarity, reactivity, phase of matter, color, magnetism, as well as the biological activity. This pair exerts repulsive forces on the bonding pairs of electrons. To form the Lewis structure of Sulfur Dioxide, we need first to determine the number of valence electrons available. Electron-pair Geometry versus Molecular Structure. Molecular geometry, also known as the molecular structure, is the three-dimensional structure or arrangement of atoms in a molecule. 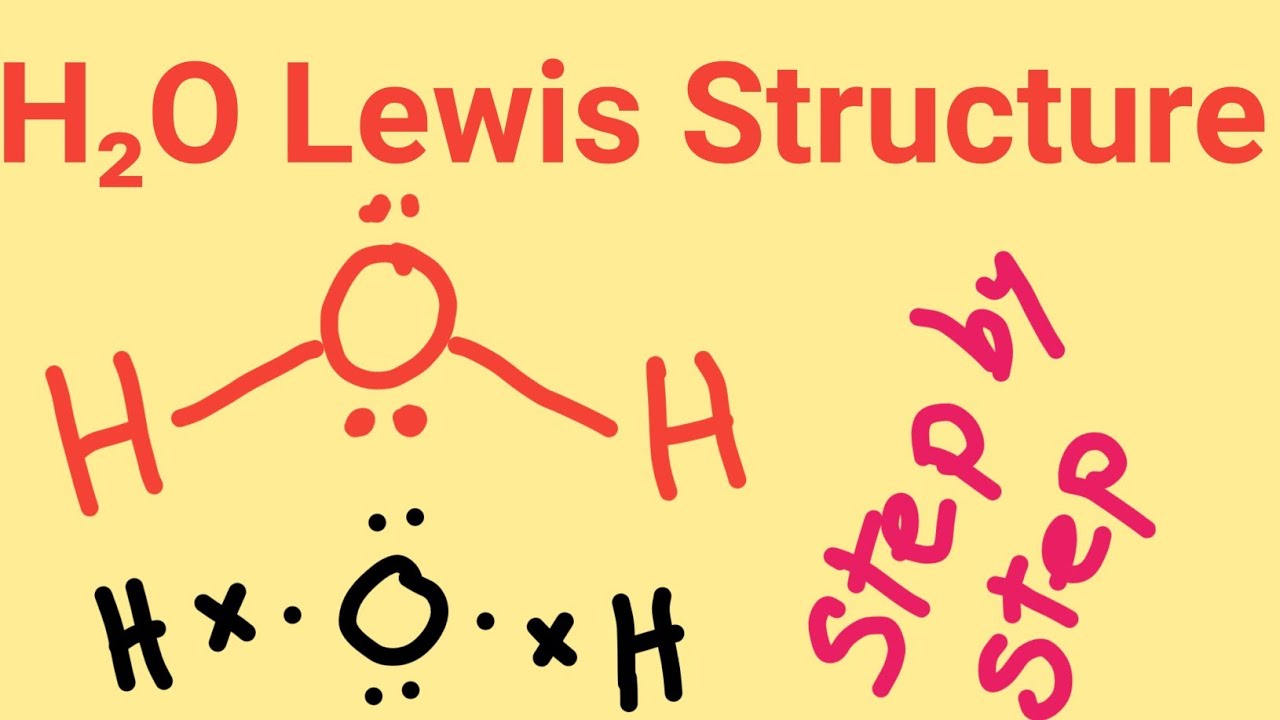
Write the correct skeletal structure for the molecule. The shape is distorted because of the lone pairs of electrons. In short, these are the steps you need to follow for drawing a Lewis structure:ġ. First, we need to draw the Lewis structure of H 2O. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
